|
2/21/2023 0 Comments Chromium iii sulfide formula Chromium III Sulfate is non combustible.It is corrosive in nature and categorized as an irritant.It is very harmful to the environment and a major health hazard.Chromium III Sulfate is an inorganic ionic compound.The molecular formula of Chromium III Sulfate is Cr 2(SO 4) 3.It serves as electrolyte to obtain chromium metal.It is used in chrome plating to decorate and protect metals.It is also used in green paints, ceramic glazes and inks.It is used in textile dyes particularly for dyeing of khaki cloth.It is extensively used in the leather industry during the process of tanning.It turns green when it is subjected to heat.Ĭhromium III Sulfate is produced as a byproduct in the process of Jones Oxidation during which a solution of sodium dichromate or potassium dichromate is dissolved with sulfuric acid to oxidize alcohols.Ĭhromium(III) sulfate can also be obtained by treating chromium III hydroxide with dilute sulfuric acid.Chromium III sulfate is a blue-gray or violet-gray amorphous solid.When we write the two together we get Cr 2 (SO 4 ) 3. So, to arrive at the formula of Chromium III Sulfate, we will interchange the charges of ions Cr +3and SO 4 -2 to get Cr 2 and (SO 4 ) 3.Read More: Conservation of Biodiversity Sample Questions Ques. It is corrosive in nature and categorized as an irritant. It is very harmful to the environment and a major health hazard. Chromium III Sulfate is an inorganic ionic compound. Cr is placed outside the parentheses as it is not a polyatomic ion. The molecular formula of Chromium III Sulfate is Cr 2 (SO 4) 3. So, polyatomic ion SO 4 -2 will be placed inside parenthesis. 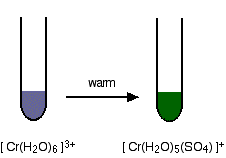
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |


 RSS Feed
RSS Feed
